|
And we see that in oxygen, it has six electrons assigned to it in an isolated atom. 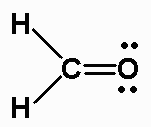
So I take half of eight and that's going to give me a formal charge of zero as well. It would have four electrons in the isolated atom. Now, I'm going to look at my carbon atom. I notice that it's going to be the same for both hydrogens, because they both have the same type of structure in this particular Lewis structure. So we’re going to put a little zero there to record that. So it has two bonding electrons around it I take half of that, and I see that I get a formal charge of zero for my hydrogen. I subtract the number of non bonding electrons assigned to it in the Lewis Structure which is zero in this case, and half of the bonding electrons. So I'm going to start with my hydrogen atom, and what I look at is the number of electrons assigned to hydrogen in an isolated atom, which would just be one electron. So now I need to look at each atom to determine the formal charge on each of these atoms. And I can use that now to find the formal charges. So now that gives me the Lewis structure for H2CO. And I have everybody with an octet, well for hydrogen it has a duet. So I'm going to redraw this structure, so I have an oxygen now with only two lone pairs of electrons, with a double bond to carbon and a single bond to hydrogen. Now I see that, while my oxygen has an octet, my carbon does not and so I need to move electrons from that oxygen atom into forming a double bond here between the carbon and the oxygen. So I've filled out my skeletal structure, I've used up six of my electrons I have six electrons remaining, so I'm going to fill in the octet on my terminal atoms so it's really only the oxygen I have to worry about the hydrogen only wants two electrons so it's perfectly fine there. Hydrogen always has to be terminal atoms, and oxygen will be a terminal atom because carbon is less electro-negative than oxygen. I see that carbon is going to be a central atom. And so I have a total of 12 electrons in my structure. For hydrogen I have two hydrogen atoms, each with one valence, plus four for the carbon, plus six for the oxygen. 
So I'm going to first count the number of valence electrons I have in the molecule. So the first thing I need to do is draw my Lewis structure, because formal charge depends on knowing how atoms are connected to one another. Determine the formal charge on atoms in H2CO.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
